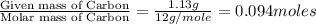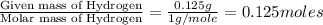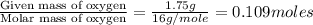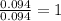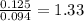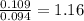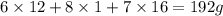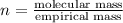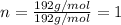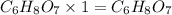A 3.00g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 192./gmol, is burned completely in excess oxygen, and the mass of the products carefully measured: product mass carbon dioxide 4.13g water 1.13g Use this information to find the molecular formula of X.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 23.06.2019 00:00
This statement about matter and its behavior is best classified as a
Answers: 1
You know the right answer?
A 3.00g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to hav...
Questions

Mathematics, 16.10.2020 08:01

History, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01


Chemistry, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01


History, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01

Chemistry, 16.10.2020 08:01

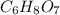
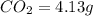
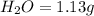
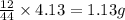 of carbon will be contained.
of carbon will be contained.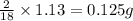 of hydrogen will be contained.
of hydrogen will be contained.