
The half-life of radium-226 is 1590 years. (a) A sample of radium-226 has a mass of 50 mg. Find a formula for the mass of the sample that remains after t years. (b) Find the mass after 500 years correct to the nearest milligram. (c) When will the mass be reduced to 40 mg

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1

Chemistry, 22.06.2019 22:30
Why is it possible for different microorganisms to extract energy not only from carbohydrates and other biological molecules but from a large variety of substances?
Answers: 1
You know the right answer?
The half-life of radium-226 is 1590 years. (a) A sample of radium-226 has a mass of 50 mg. Find a fo...
Questions



Chemistry, 29.07.2019 23:50


Mathematics, 29.07.2019 23:50


Mathematics, 29.07.2019 23:50

Arts, 29.07.2019 23:50

Mathematics, 29.07.2019 23:50

English, 29.07.2019 23:50


Mathematics, 29.07.2019 23:50

Mathematics, 29.07.2019 23:50




Social Studies, 29.07.2019 23:50

Social Studies, 29.07.2019 23:50

Mathematics, 29.07.2019 23:50

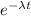
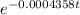
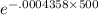
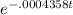
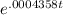 = 1.25
= 1.25 

