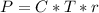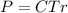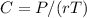Chemistry, 11.06.2020 16:02 bethdove9466
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute temperature and the gas constant . Suppose the osmotic pressure of a certain solution is measured to be at an absolute temperature o of 312. K. Write an equation that will let you calculate the molarity c of this solution.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1


You know the right answer?
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute tempe...
Questions

Mathematics, 19.03.2020 03:00

Mathematics, 19.03.2020 03:00

Mathematics, 19.03.2020 03:00

Mathematics, 19.03.2020 03:00












Health, 19.03.2020 03:01


Chemistry, 19.03.2020 03:01


Mathematics, 19.03.2020 03:01