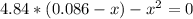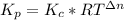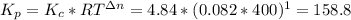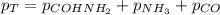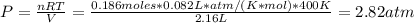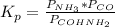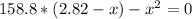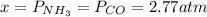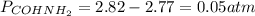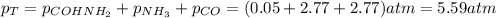Chemistry, 11.06.2020 02:57 dontworry48
Formamide decomposes at high temperature. If 0.186 mol of formamide (HCONH2) dissociates in a 2.16 L flask at 400 K, what are the concentrations of all species present at equilibrium at 400 K? (hint: calculate concentrations first) (b) What is the total pressure in the container at equilibrium?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1
You know the right answer?
Formamide decomposes at high temperature. If 0.186 mol of formamide (HCONH2) dissociates in a 2.16 L...
Questions



English, 29.08.2019 04:00


English, 29.08.2019 04:00






History, 29.08.2019 04:00

Biology, 29.08.2019 04:00

English, 29.08.2019 04:00

Social Studies, 29.08.2019 04:00

Mathematics, 29.08.2019 04:00

Arts, 29.08.2019 04:00


English, 29.08.2019 04:00

Mathematics, 29.08.2019 04:00

History, 29.08.2019 04:00

![K_{c} = \frac{[NH_{3}][CO]}{[COHNH_{2}]} = 4.84 (400 K)](/tpl/images/0682/5560/28fef.png)
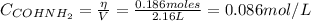
![K_{c} = \frac{[NH_{3}][CO]}{[COHNH_{2}]} = \frac{x*x}{0.086 - x}](/tpl/images/0682/5560/05a27.png)