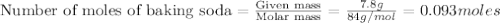Chemistry, 10.06.2020 06:57 smithsa10630
1.)A strong acid solution requires 3.2 grams of sulfuric acid (H2SO4). How many molecules of sulfuric acid are in the solution? 2.) While measuring out the sulfuric acid you accidentally spilled some of it! Before trying to clean it up you put some baking soda (NaHCO3) on to it neutralize it. If you scatter 7.8 g of baking soda on the acid how many moles of baking soda have you used?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 07:00
Which set of characteristics best describes igneous rock? a) largest type of rock, made of organic matter, hardest type of rock b) least abundant type of rock, made of other rocks, made mostly of minerals c) found on all continents, contains wavy bands of stripes, contains fossils d) most abundant type in earth's crust, made of magma/lava, contains no fossils
Answers: 1


Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1
You know the right answer?
1.)A strong acid solution requires 3.2 grams of sulfuric acid (H2SO4). How many molecules of sulfuri...
Questions

Mathematics, 03.11.2020 03:20

Mathematics, 03.11.2020 03:20



Social Studies, 03.11.2020 03:20


Mathematics, 03.11.2020 03:20

Mathematics, 03.11.2020 03:20



History, 03.11.2020 03:20


Mathematics, 03.11.2020 03:20



Biology, 03.11.2020 03:20




Social Studies, 03.11.2020 03:20

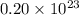 molecules of sulfuric acid in the solution.
molecules of sulfuric acid in the solution.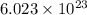 of particles.
of particles.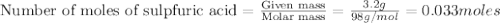
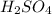 contains =
contains = 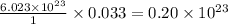 molecules of sulfuric acid
molecules of sulfuric acid