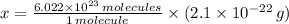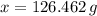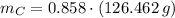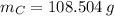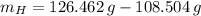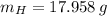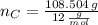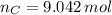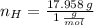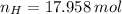Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2
You know the right answer?
Un hidrocarburo tiene como composición en masa: C= 85.8% ; H= 14.2% . Como dato nos brindan que una...
Questions


Mathematics, 31.10.2019 09:31

English, 31.10.2019 09:31

Mathematics, 31.10.2019 09:31




Mathematics, 31.10.2019 09:31


History, 31.10.2019 09:31



Mathematics, 31.10.2019 09:31

Health, 31.10.2019 09:31



Biology, 31.10.2019 09:31


Mathematics, 31.10.2019 09:31

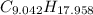
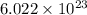 moléculas. La masa de un mol se determina mediante regla de tres simple:
moléculas. La masa de un mol se determina mediante regla de tres simple: