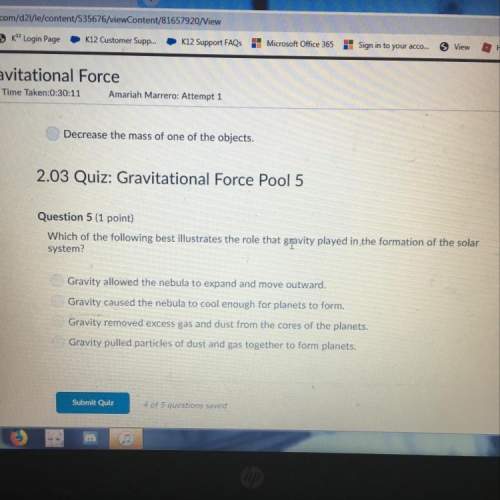
Chemistry, 06.06.2020 14:57 restinpeicejahseh
Consider The reaction Cu2O(s) + C (s) > 2 Cu (s) + CO (g)
When 11,5 g of C are allowed to react with 114,5 g of Cu2O, 87,4 g of Cu are obtained. Find
the limiting reactant,

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
How many atoms of oxygen are contained in 160 grams of n2o3
Answers: 2

Chemistry, 22.06.2019 01:40
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
Consider The reaction Cu2O(s) + C (s) > 2 Cu (s) + CO (g)
When 11,5 g of C are allowed to react...
Questions

Mathematics, 17.02.2021 18:50


Chemistry, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50

English, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50

English, 17.02.2021 18:50

English, 17.02.2021 18:50



Mathematics, 17.02.2021 18:50


Mathematics, 17.02.2021 18:50

History, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50


History, 17.02.2021 19:00

Mathematics, 17.02.2021 19:00

Computers and Technology, 17.02.2021 19:00