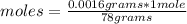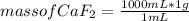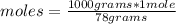In the background information, it was stated that CaF2 has solubility, at room temperature, of 0.00160 g per 100 g of water. How many moles of CaF2 can dissolve in 100 g of water? If the density of a saturated solution of CaF2 is 1.00 g/mL, how many moles of CaF2 will dissolve in exactly 1.00 L of solution?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
In the background information, it was stated that CaF2 has solubility, at room temperature, of 0.001...
Questions



Chemistry, 27.12.2019 17:31





Social Studies, 27.12.2019 17:31




Mathematics, 27.12.2019 17:31

English, 27.12.2019 17:31

Mathematics, 27.12.2019 17:31

History, 27.12.2019 17:31



Mathematics, 27.12.2019 17:31

History, 27.12.2019 17:31

Mathematics, 27.12.2019 17:31