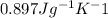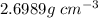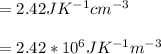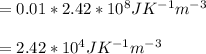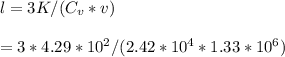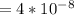Calculate the mean free path of electrons in a metal, such as silver, at room temperature form heat capacity and heat conduction measurements. Take EF ¼ 5 eV, K ¼ 4:29 102 J/s m K, and Cel v ¼ 1% of the lattice heat capacity. (Hint: Remember that the heat capacity in (21.8) is given per unit volume!)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 11:00
An object becomes electrically charged when: electrons are created in it electrons from it are destroyed electrons are transferred to it protons from it are destroyed protons are created in it
Answers: 1

You know the right answer?
Calculate the mean free path of electrons in a metal, such as silver, at room temperature form heat...
Questions


Mathematics, 28.10.2019 00:43

Biology, 28.10.2019 00:43

Mathematics, 28.10.2019 00:43




History, 28.10.2019 00:43



Mathematics, 28.10.2019 00:43






English, 28.10.2019 00:43


Mathematics, 28.10.2019 00:43

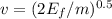
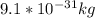 and
and 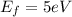 we get
we get 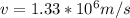
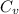 is 1% of lattice heat capacity
is 1% of lattice heat capacity