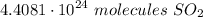Chemistry, 03.06.2020 13:07 janeou17xn
How many molecules are in 7.32 moles of sulfur dioxide?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:20
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1

Chemistry, 23.06.2019 06:00
Robert leaves a chocolate bar in his car while attending school all day. when he goes to his car in the afternoon, the bat has changed into gooey liquid. what happened to the chocolate bar
Answers: 1

Chemistry, 23.06.2019 07:00
What are the trends and exceptions to the trends in electron affinity?
Answers: 1

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2
You know the right answer?
How many molecules are in 7.32 moles of sulfur dioxide?...
Questions


Computers and Technology, 22.08.2019 20:30


Computers and Technology, 22.08.2019 20:30







History, 22.08.2019 20:30


Chemistry, 22.08.2019 20:30


Computers and Technology, 22.08.2019 20:30

English, 22.08.2019 20:30

English, 22.08.2019 20:30


History, 22.08.2019 20:30

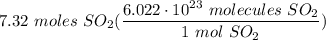 [DA] Multiply [Cancel out units]:
[DA] Multiply [Cancel out units]: