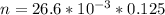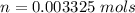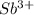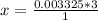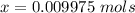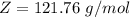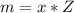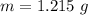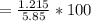Chemistry, 03.06.2020 02:59 Queenhagar
The quantity of antimony in a sample can be determined by an oxidation–reduction titration with an oxidizing agent.
A 5.85 g sample of stibnite, an ore of antimony, is dissolved in hot, concentrated HCl(aq) and passed over a reducing agent so that all the antimony is in the form Sb3+(aq). The Sb3+(aq) is completely oxidized by 26.6 mL of a 0.125 M aqueous solution of KBrO3(aq). The unbalanced equation for the reaction is:
BrO3-(aq) + Sb3+(aq) > Br-(aq) + Sb5+(aq) (unbalanced)
(A) Calculate the amount of antimony in the sample and its percentage in the ore.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Abullet found at a crime scene may be used as evidence in a trial if the percentage of metals match to the composition of metals in a bullet from the suspect's ammunition. a forensic scientist's analysis of the bullet shows that it contains 11.9 g of lead, 0.5 g of tin, and 0.8 b of antimony. what is the percentage of lead metal in the bullet? express your answers to the one's place.
Answers: 2

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
The quantity of antimony in a sample can be determined by an oxidation–reduction titration with an o...
Questions

History, 29.01.2021 20:20

Mathematics, 29.01.2021 20:20

Mathematics, 29.01.2021 20:20


Spanish, 29.01.2021 20:20



English, 29.01.2021 20:20

Mathematics, 29.01.2021 20:20



Mathematics, 29.01.2021 20:20

Mathematics, 29.01.2021 20:20

Mathematics, 29.01.2021 20:20


Health, 29.01.2021 20:20

English, 29.01.2021 20:20

Biology, 29.01.2021 20:20


English, 29.01.2021 20:20

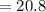 %
%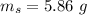
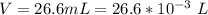
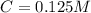
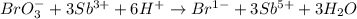
 is
is