N2(g) + 3H2(g) <-> 2NH3(g) + energy

Chemistry, 02.06.2020 19:58 roxymiller3942
Given the equation for a system at equilibrium:
N2(g) + 3H2(g) <-> 2NH3(g) + energy
If only the concentration of N2(g) is increased, the concentration of
1. NH3(g) increases
2. NH3(g) remains the same
3. H2(g) increases
4. H2(g) remains the same

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1

Chemistry, 23.06.2019 04:31
Use the drop-down menus to label each of the following changes p for physical change and c for chemical change. the substance changes to a new substance. the original substance can be recovered. the color changes. gas is produced and given off. the substance changes size, shape, or volume.
Answers: 2
You know the right answer?
Given the equation for a system at equilibrium:
N2(g) + 3H2(g) <-> 2NH3(g) + energy
N2(g) + 3H2(g) <-> 2NH3(g) + energy
Questions

Mathematics, 06.01.2021 04:10


Chemistry, 06.01.2021 04:10

Spanish, 06.01.2021 04:10





Mathematics, 06.01.2021 04:10




Social Studies, 06.01.2021 04:10



Mathematics, 06.01.2021 04:10



Mathematics, 06.01.2021 04:10

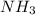 increases.
increases.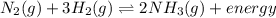
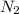 is increased , according to the Le-Chatlier's principle, the reaction has to shift to right or forward direction. In order to do that the concentration of products has to increase.
is increased , according to the Le-Chatlier's principle, the reaction has to shift to right or forward direction. In order to do that the concentration of products has to increase.

