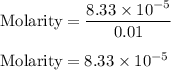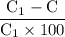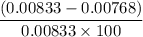You weighed out 0.020 g of your crude aspirin product in order to determine the amount of salicylic acid impurity. Following the procedure outlined in the manual, you dissolved the solid and diluted the solution to a final volume of 10.0 mL. If the absorbance of your sample solution was 1.07, what was the percent salicylic acid in your product

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2
You know the right answer?
You weighed out 0.020 g of your crude aspirin product in order to determine the amount of salicylic...
Questions


Chemistry, 11.03.2021 14:00

Arts, 11.03.2021 14:00




Physics, 11.03.2021 14:00

Social Studies, 11.03.2021 14:00

Biology, 11.03.2021 14:00

Chemistry, 11.03.2021 14:00

English, 11.03.2021 14:00


Mathematics, 11.03.2021 14:00

Mathematics, 11.03.2021 14:00


Mathematics, 11.03.2021 14:00

Physics, 11.03.2021 14:00


Computers and Technology, 11.03.2021 14:00

English, 11.03.2021 14:00

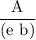
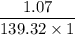 ( ε = 139.32)
( ε = 139.32)