Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 01:30
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1

Chemistry, 23.06.2019 01:40
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1

Chemistry, 23.06.2019 05:00
Match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) amount of product predicted to be produced by the given reactants theoretical yield c) reactant that can produce more of the product
Answers: 3

Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3
You know the right answer?
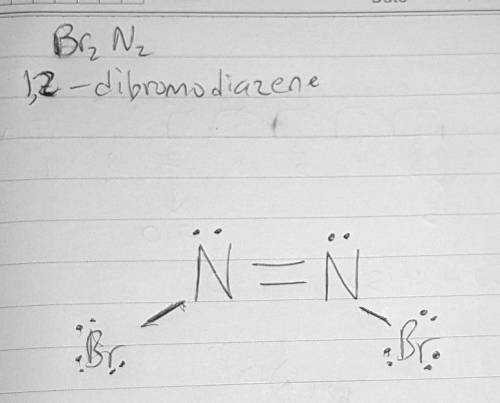
Lewis diagram for Br2N2...
Questions


Mathematics, 17.07.2019 00:00

English, 17.07.2019 00:00

History, 17.07.2019 00:00


Physics, 17.07.2019 00:00

Social Studies, 17.07.2019 00:00



History, 17.07.2019 00:00

Mathematics, 17.07.2019 00:00

History, 17.07.2019 00:00


Business, 17.07.2019 00:00




Physics, 17.07.2019 00:00

Physics, 17.07.2019 00:00

Social Studies, 17.07.2019 00:00