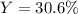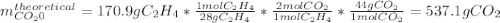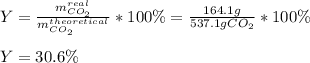Chemistry, 29.05.2020 20:02 kargarzadehsm
In an experiment, 170.9 g of C2H4 was reacted with an excess of O2, 164.1 g of CO2 is produced.
C2H4 (g) + 3 O2(g) → 2 CO2 (g) + 2 H2O (l)
What is the percent yield of this reaction?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
If the particles in a sample of matter have an orderly arrangement and move only in place, the sample is a
Answers: 1


Chemistry, 22.06.2019 01:30
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

You know the right answer?
In an experiment, 170.9 g of C2H4 was reacted with an excess of O2, 164.1 g of CO2 is produced.
Questions

Mathematics, 05.05.2020 07:35










Mathematics, 05.05.2020 07:35

Mathematics, 05.05.2020 07:35




Mathematics, 05.05.2020 07:35


Mathematics, 05.05.2020 07:35

Biology, 05.05.2020 07:36