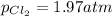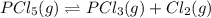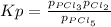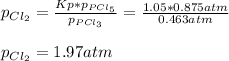Chemistry, 27.05.2020 23:06 hillarytrinh
3. The equilibrium constant KP for the decomposition of phosphorus pentachloride to phosphorus trichloride and molecular chlorine according to the chemical equation given below is found to be 1.05 at 250 °C. If the equilibrium partial pressures of PCl5 and PCl3 are 0.875 atm and 0.463 atm, respectively, what is the equilibrium partial pressure of Cl2 at 250 °C? PCl5(g) PCl3(g) + Cl2(g)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:50
Aluminum–lithium (al-li) alloys have been developed by the aircraft industry to reduce the weight and improve the performance of its aircraft. a commercial aircraft skin material having a density of 2.47 g/cm3 is desired. compute the concentration of li (in wt%) that is required.
Answers: 3

Chemistry, 22.06.2019 21:30
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2


Chemistry, 23.06.2019 09:00
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate according to the following equation:
Answers: 2
You know the right answer?
3. The equilibrium constant KP for the decomposition of phosphorus pentachloride to phosphorus trich...
Questions


History, 16.09.2019 19:00


Mathematics, 16.09.2019 19:00




History, 16.09.2019 19:00

History, 16.09.2019 19:00


Physics, 16.09.2019 19:00




Mathematics, 16.09.2019 19:00


Mathematics, 16.09.2019 19:00