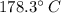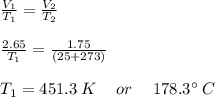Chemistry, 25.05.2020 22:57 darianhaynes
An ideal gas in a sealed container has an initial volume of 2.65 L. At constant pressure, it is cooled to 25.00 ∘C, where its final volume is 1.75 L. What was the initial temperature?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1


Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1
You know the right answer?
An ideal gas in a sealed container has an initial volume of 2.65 L. At constant pressure, it is cool...
Questions


Mathematics, 29.11.2020 22:30




Mathematics, 29.11.2020 22:30





History, 29.11.2020 22:30



Arts, 29.11.2020 22:30



Biology, 29.11.2020 22:30

Mathematics, 29.11.2020 22:30

Biology, 29.11.2020 22:30

Mathematics, 29.11.2020 22:30