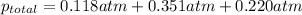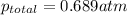Chemistry, 25.05.2020 05:57 lamooothegoat
What is the total pressure in atmospheres of a mixture of three gases with partial pressures of 0.118 atm, 35.6 kPa, and 167 mm Hg?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
What must happen before a body cell can begin mitotic cell division
Answers: 2

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3
You know the right answer?
What is the total pressure in atmospheres of a mixture of three gases with partial pressures of 0.11...
Questions

Mathematics, 01.07.2020 20:01


Mathematics, 01.07.2020 20:01



Mathematics, 01.07.2020 20:01








Mathematics, 01.07.2020 20:01



Mathematics, 01.07.2020 20:01

Computers and Technology, 01.07.2020 20:01


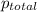 =total pressure of gases = ?
=total pressure of gases = ?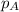 = partial pressure of A = 0.118 atm
= partial pressure of A = 0.118 atm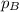 = partial pressure of B = 35.6 kPa = 0.351 atm (1kPa =0.0098atm)
= partial pressure of B = 35.6 kPa = 0.351 atm (1kPa =0.0098atm)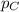 = partial pressure of C = 167 mm Hg = 0.220 atm (1mmHg=0.0013 atm)
= partial pressure of C = 167 mm Hg = 0.220 atm (1mmHg=0.0013 atm)