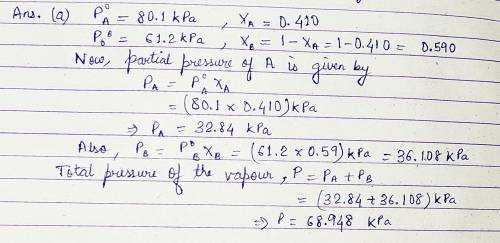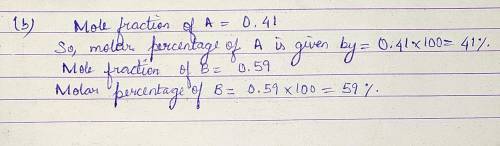Someone pls help me, I need the answer ASAP!!
Pure A (liq) has vapour pressure at 421 K is 80....

Chemistry, 24.05.2020 17:57 Chavens520
Someone pls help me, I need the answer ASAP!!
Pure A (liq) has vapour pressure at 421 K is 80.1 kPa, and pure B (liq) is 61.2 kPa. The two substance form ideal liquid and gaseous mixtures. If the equilibrium composition of a mixture is established, in which the mole fraction of A in the vapour is 0.410, Calculate:
(a) The total pressure of the vapour.
(b) The composition of the liquid mixture.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Theoretically, which metal should be the most reactive? hydrogen lithium francium fluorine
Answers: 1

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1


Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Questions






English, 31.10.2019 00:31



Biology, 31.10.2019 00:31



Biology, 31.10.2019 00:31





Mathematics, 31.10.2019 00:31

Spanish, 31.10.2019 00:31

History, 31.10.2019 00:31

Mathematics, 31.10.2019 00:31