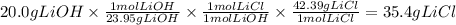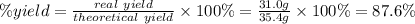Chemistry, 19.05.2020 02:15 jluckie080117
A reaction starts with 20.0 g of lithium hydroxide (LiOH) and actually produces 31.0 g of lithium chloride (LiCl), what is the percent yield? (Hint: First calculate the theoretical yield of lithium chloride (LiCl))
64.5%
88.6%
81.5%
92.8%


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1


Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 03:50
How many moles of potassium are needed to react completely with 12.8 moles of magnessium bromide?
Answers: 2
You know the right answer?
A reaction starts with 20.0 g of lithium hydroxide (LiOH) and actually produces 31.0 g of lithium ch...
Questions


Social Studies, 04.09.2020 23:01

Mathematics, 04.09.2020 23:01



Social Studies, 04.09.2020 23:01


Computers and Technology, 04.09.2020 23:01

Mathematics, 04.09.2020 23:01



English, 04.09.2020 23:01

Mathematics, 04.09.2020 23:01



Advanced Placement (AP), 04.09.2020 23:01


Computers and Technology, 04.09.2020 23:01


Biology, 04.09.2020 23:01