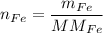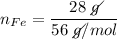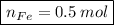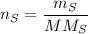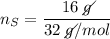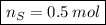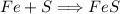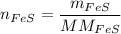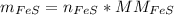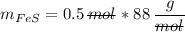Chemistry, 25.08.2019 06:00 malikbryant2002
In a chemical reaction, 28g of iron reacts with 16g of sulfur to produce of iron sulfide.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 01:30
Concentrations expressed as a percent by mass are useful when the solute is a a. liquid b. gas c. solid
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3
You know the right answer?
In a chemical reaction, 28g of iron reacts with 16g of sulfur to produce of iron sulfide....
Questions


Business, 19.11.2019 22:31

Social Studies, 19.11.2019 22:31

Mathematics, 19.11.2019 22:31


Chemistry, 19.11.2019 22:31

History, 19.11.2019 22:31



Mathematics, 19.11.2019 22:31

Mathematics, 19.11.2019 22:31

Social Studies, 19.11.2019 22:31

English, 19.11.2019 22:31


Biology, 19.11.2019 23:31

English, 19.11.2019 23:31




Mathematics, 19.11.2019 23:31