
Chemistry, 06.05.2020 20:04 MikeWrice4494
Mercury(II) oxide (HgO) decomposes to form mercury (Hg) and oxygen (O2). The balanced chemical equation is shown below.2HgO Right arrow. 2Hg + O2The molar mass of HgO is 216.59 g/mol. The molar mass of O2 is 32.00 g/mol. How many moles of HgO are needed to produce 250.0 g of O2?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 11:00
An object becomes electrically charged when: electrons are created in it electrons from it are destroyed electrons are transferred to it protons from it are destroyed protons are created in it
Answers: 1

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2

Chemistry, 22.06.2019 21:00
Which property of water causes water drops to bead on a freshly waxed car?
Answers: 2
You know the right answer?
Mercury(II) oxide (HgO) decomposes to form mercury (Hg) and oxygen (O2). The balanced chemical equat...
Questions

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

English, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

History, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

English, 18.09.2020 21:01

Chemistry, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Social Studies, 18.09.2020 21:01

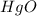 are needed to produce 250.0 g of
are needed to produce 250.0 g of 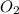
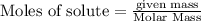
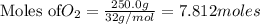
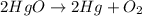
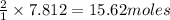 of
of 

