
Chemistry, 06.05.2020 06:44 blackchina71
If 20.5 g of chlorine is reacted with 20.5 g of sodium, which reactant is in excess?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2
You know the right answer?
If 20.5 g of chlorine is reacted with 20.5 g of sodium, which reactant is in excess?...
Questions

Advanced Placement (AP), 20.09.2021 05:00

Mathematics, 20.09.2021 05:00

Mathematics, 20.09.2021 05:00



Chemistry, 20.09.2021 05:00

Mathematics, 20.09.2021 05:00


Mathematics, 20.09.2021 05:00

Chemistry, 20.09.2021 05:00

English, 20.09.2021 05:00

Computers and Technology, 20.09.2021 05:00


Computers and Technology, 20.09.2021 05:00



History, 20.09.2021 05:00

Business, 20.09.2021 05:00


Business, 20.09.2021 05:00

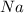 is in excess.
is in excess.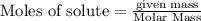
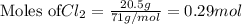

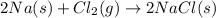
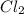 require 2 moles of
require 2 moles of 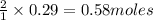 of
of 

