
Chemistry, 06.05.2020 03:32 RealSavage4Life
Consider these generic half-reactions. Half-reaction E° (V) X+(aq)+e−⟶X(s) 1.52 Y2+(aq)+2e−⟶Y(s) −1.17 Z3+(aq)+3e−⟶Z(s) 0.84 Identify the strongest oxidizing agent. X+ Y2+ X Z Y Z3+ Identify the weakest oxidizing agent. X Z3+ Y Y2+ Z X+ Identify the strongest reducing agent. Z3+ X+ Y2+ Z X Y Identify the weakest reducing agent. Y Z X+ X Y2+ Z3+ Which substances can oxidize Z ?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1


Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1
You know the right answer?
Consider these generic half-reactions. Half-reaction E° (V) X+(aq)+e−⟶X(s) 1.52 Y2+(aq)+2e−⟶Y(s) −1....
Questions

Mathematics, 02.01.2021 03:00



Computers and Technology, 02.01.2021 03:00



Mathematics, 02.01.2021 03:00

Mathematics, 02.01.2021 03:00

Mathematics, 02.01.2021 03:00

Arts, 02.01.2021 03:00

English, 02.01.2021 03:00

Mathematics, 02.01.2021 03:00





Mathematics, 02.01.2021 03:00

Chemistry, 02.01.2021 03:00

Computers and Technology, 02.01.2021 03:00

Chemistry, 02.01.2021 03:00

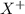
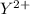
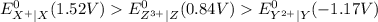
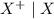 is higher than the reduction potential of the half cell
is higher than the reduction potential of the half cell 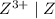 therefore
therefore 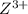 and itself gets converted into X.
and itself gets converted into X.

