
Chemistry, 06.05.2020 01:35 cbelew0001ouje4i
Which of the following item(s) explain the differences between the Ka values. Choose one or more: A. The oxidation state for oxygen in trifluoroacetate is more negative than the oxidation state for oxygen in acetate. B. The trifluoroacetate molecule has more resonance structures than the acetate molecule. C. The electron-withdrawing fluorine atoms pull electron density from the oxygen in trifluoroacetate. The negative charge is more stabilized in trifluoroacetate by this effect. D. The negative charge is on the more electronegative fluorine atom in trifluoroacetate.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:00
The study of witch tree monkeys feed in is part of the science life
Answers: 1

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
Which of the following item(s) explain the differences between the Ka values. Choose one or more: A....
Questions

Social Studies, 27.12.2021 18:00

Business, 27.12.2021 18:00

Computers and Technology, 27.12.2021 18:00

History, 27.12.2021 18:00

SAT, 27.12.2021 18:00

Mathematics, 27.12.2021 18:00

Physics, 27.12.2021 18:00



Mathematics, 27.12.2021 18:10



History, 27.12.2021 18:10

French, 27.12.2021 18:10


English, 27.12.2021 18:10


English, 27.12.2021 18:10


Social Studies, 27.12.2021 18:10

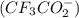 ,The acetate anion Two oxygen atoms.
,The acetate anion Two oxygen atoms.


