
Calculate Z and V for ethylene at 25°C and 12 bar by the following equations: (a) The truncated virial equation [Eq. (3.38)] with the following experimental values of virial coefficients: B = −140 cm3·mol−1 C = 7200 cm6·mol−2 (b) The truncated virial equation [Eq. (3.36)], with a value of B from the generalized Pitzer correlation [Eqs. (3.58)–(3.62)] (c) The Redlich/Kwong equation (d) The Soave/Redlich/Kwong equation (e) The Peng/Robinson equation

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Write two balanced equations 1. dissolving of solid sodium hydroxide in water 2. the reaction of sodium hydroxide solution with hydrochloric acid
Answers: 1

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 19:50
What is the wavelength of a wave with a velocity of 50 m/s and a frequency of 5hz a 250 m b 0.1 m c 10m d 0.01 m
Answers: 2

Chemistry, 23.06.2019 02:30
Ascientist wants to know how individual lions within a pride interact with each other in their own environment. to do this, the scientist sedates and tags all of the lions within a pride. then, he places several remotely-controlled video cameras near the lions' den and performs an observational field study. he collects continuous video footage over the span of one year, analyzes the video, and then forms conclusions based on his observations.
Answers: 2
You know the right answer?
Calculate Z and V for ethylene at 25°C and 12 bar by the following equations: (a) The truncated viri...
Questions


Health, 25.12.2021 18:40


Chemistry, 25.12.2021 18:50







Social Studies, 25.12.2021 18:50



Business, 25.12.2021 18:50





SAT, 25.12.2021 18:50

Mathematics, 25.12.2021 18:50

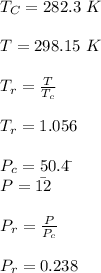
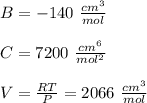
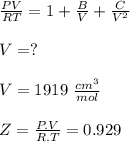
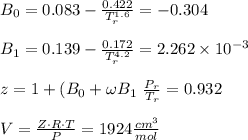
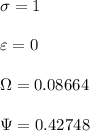
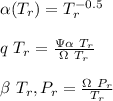
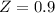
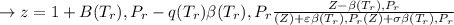
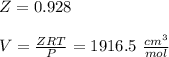
![\sigma =1\\\\\varepsilon = 0\\\\ \Omega= 0.08664\\\\ \Psi= 0.42748 \\\\\alpha (T_r) \omega =[ 1+ 0.480 + 1.574 \omega -0.176 \omega^2 (1-T_r^{\frac{1}{2}})]^2\\\\ \alpha (T_r)=\frac{\Psi \alpha (T_r) , \omega}{\Omega \ T_r}\\\\\beta (T_r), P_r=\frac{\omega\ P_r}{T_r}\\\\](/tpl/images/0640/3828/ecc5c.png)
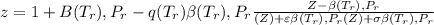
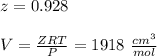
![\sigma =1+\sqrt{2}\\\\\varepsilon = 1-\sqrt{2}\\\\ \Omega= 0.07779\\\\ \Psi= 0.45724 \\\\\alpha (T_r) \omega =[ 1+ 0.37464 + 1.5422 \omega -0.26992 \omega^2 (1-T_r^{\frac{1}{2}})]^2\\\\ \alpha (T_r)=\frac{\Psi \alpha (T_r) , \omega}{\Omega \ T_r}\\\\\beta (T_r), P_r=\frac{\omega\ P_r}{T_r}\\\\](/tpl/images/0640/3828/e71f8.png)




