Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:20
Amixture of gaseous sulfur dioxide and oxygen are added to a reaction vessel and heated to 1000 k where they react to form so3(g). if the vessel contains 0.669 atm so2(g), 0.395 atm o2(g), and 0.0851 atm so3(g) after the system has reached equilibrium, what is the equilibrium constant kp for the reaction: 2 so2(g) o2(g) ⇌ 2 so3(g)
Answers: 3

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1


You know the right answer?
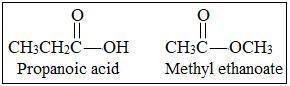
Which formula represents an isomer of ch3-ch2-cooh...
Questions


Computers and Technology, 16.12.2021 14:20

English, 16.12.2021 14:20

History, 16.12.2021 14:20

English, 16.12.2021 14:20



History, 16.12.2021 14:20


History, 16.12.2021 14:20



Physics, 16.12.2021 14:20

Social Studies, 16.12.2021 14:20

Computers and Technology, 16.12.2021 14:20

Computers and Technology, 16.12.2021 14:20

History, 16.12.2021 14:20

Mathematics, 16.12.2021 14:20

English, 16.12.2021 14:20

English, 16.12.2021 14:30