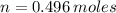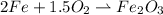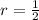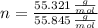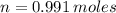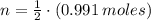Chemistry, 05.05.2020 00:28 bricksaspares
Iron and oxygen form rust. How many moles of rust should be produced if 1
55.321 grams of iron reacted? *

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2
You know the right answer?
Iron and oxygen form rust. How many moles of rust should be produced if 1
55.321 grams of iron...
55.321 grams of iron...
Questions

Chemistry, 26.06.2020 17:01





Business, 26.06.2020 17:01




English, 26.06.2020 17:01

Mathematics, 26.06.2020 17:01


Computers and Technology, 26.06.2020 17:01