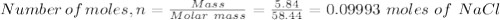A student has a mixture of salt (NaCl) and sugar (C12H22O11). To determine the percentage, the student measures out 5.84 grams of the mixture and dissolves this in 100.0 mL of water. The student knows that aqueous salt with aqueous silver nitrate (AgNO3) to form solid silver chloride (AgCl). The student determines that sugar will not react with silver nitrate. How many mL of 1.0 M AgNO3 would be required to precipitate 5.84 grams of NaCl?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 22.06.2019 00:30
Which best describes why nh4+ can form an ionic bond with ci-?
Answers: 1

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2
You know the right answer?
A student has a mixture of salt (NaCl) and sugar (C12H22O11). To determine the percentage, the stude...
Questions

Social Studies, 25.10.2019 07:43

Biology, 25.10.2019 07:43

Chemistry, 25.10.2019 07:43

Mathematics, 25.10.2019 07:43


Mathematics, 25.10.2019 07:43


Mathematics, 25.10.2019 07:43


Mathematics, 25.10.2019 07:43

Mathematics, 25.10.2019 07:43

Mathematics, 25.10.2019 07:43

Mathematics, 25.10.2019 07:43


History, 25.10.2019 07:43


Social Studies, 25.10.2019 07:43

Mathematics, 25.10.2019 07:43

Mathematics, 25.10.2019 07:43