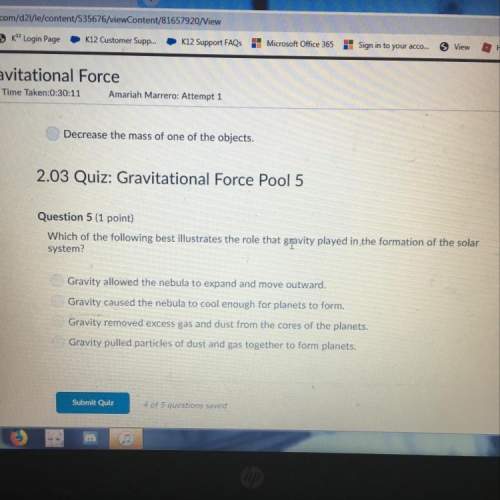
Equilibrium constant, Keq or Kc, has no units.
True
False
Reversibl...

Equilibrium constant, Keq or Kc, has no units.
True
False
Reversible reactions proceed in Only one direction.
True
False
Irreversible reactions proceed in Only one direction.
True
False
At equilibrium,
Rate of Forward reaction = Rate of reverse reaction.
True
False
Any Reversible reaction in a CLOSED SYSTEM, will reach equilibrium eventually.
True
False
Chemical Equilibrium is DYNAMIC.
True
False
In a Reversible Reaction, if the Forward reaction is ENDOTHERMIC. Then, the REVERSE ( Backward) reaction will be .
Cannot be predicted
Exothermic
Endothermic

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3


Chemistry, 23.06.2019 03:30
27 drag each label to the correct location on the image. a particular exosolar system has five planets in total: a, b, c, d, and e. the table lists the orbital periods of these planets in days. planet orbital period (days) a 600 b 80 c 1,000 d 500 e 100 move each planet to its orbit in the system.
Answers: 3
You know the right answer?
Questions



Mathematics, 01.06.2020 19:59



Mathematics, 01.06.2020 19:59






Mathematics, 01.06.2020 20:00

Physics, 01.06.2020 20:00

Mathematics, 01.06.2020 20:00

Social Studies, 01.06.2020 20:00

Mathematics, 01.06.2020 20:00

Mathematics, 01.06.2020 20:00



History, 01.06.2020 20:00