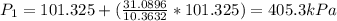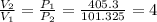Chemistry, 05.05.2020 04:21 smithmalyk4
A scuba diver that ascends to the surface too quickly can experience decompression sickness, which occurs when nitrogen that dissolves in the blood under high pressure, forms bubbles as the pressure decreases during the ascent. Therefore, an understanding of the gas laws is an important part of a scuba diver's training. In fresh water, the pressure increases by 1 atm every 34 ft below the water surface a diver descends.
If a diver ascends quickly to the surface from a depth of 102 ft without exhaling, by what factor will the volume of the diver's lungs change upon arrival at the surface?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 13:30
Ants live on acacia trees in south america. the ants feed on sugars secreted by the trees. the trees provide room for the ants to live. the ants sting any other insect or animal that comes to eat the trees. what type of relationship is this?
Answers: 1

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 19:00
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3
You know the right answer?
A scuba diver that ascends to the surface too quickly can experience decompression sickness, which o...
Questions

Mathematics, 26.01.2020 11:31


Social Studies, 26.01.2020 11:31

English, 26.01.2020 11:31

Mathematics, 26.01.2020 11:31

Biology, 26.01.2020 11:31

Mathematics, 26.01.2020 11:31


Chemistry, 26.01.2020 11:31




Social Studies, 26.01.2020 11:31



Mathematics, 26.01.2020 11:31

History, 26.01.2020 11:31



Mathematics, 26.01.2020 11:31