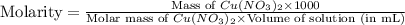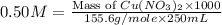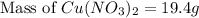Which calculation below represents the mass of
copper(II) nitrite (MM = 155.6 g/mol) should be...

Chemistry, 05.05.2020 04:15 barnettboy76
Which calculation below represents the mass of
copper(II) nitrite (MM = 155.6 g/mol) should be
weighed out to produce 250 ml of a 0.50 M solution?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Write the symbol for every chemical element that has atomic number greater than 3 and atomic mass less than 12.0 u.
Answers: 1

Chemistry, 21.06.2019 23:30
Write a paragraph that provides examples of each stage of volcanic activity, a description of the volcano, and facts about each stage.
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
Questions

Biology, 25.10.2020 21:50

Mathematics, 25.10.2020 21:50

Mathematics, 25.10.2020 21:50

Advanced Placement (AP), 25.10.2020 21:50

Mathematics, 25.10.2020 21:50

English, 25.10.2020 21:50

Mathematics, 25.10.2020 21:50

Mathematics, 25.10.2020 21:50

French, 25.10.2020 21:50

English, 25.10.2020 21:50






Mathematics, 25.10.2020 21:50

Spanish, 25.10.2020 21:50

English, 25.10.2020 21:50


Physics, 25.10.2020 21:50

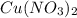 in the solution is, 19.4 grams.
in the solution is, 19.4 grams.