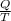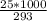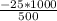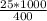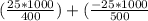Chemistry, 05.05.2020 05:40 olejlund8073
An object at 20∘C absorbs 25.0 J of heat. What is the change in entropy ΔS of the object? Express your answer numerically in joules per kelvin. ΔS = nothing J/K Request Answer Part E An object at 500 K dissipates 25.0 kJ of heat into the surroundings. What is the change in entropy ΔS of the object? Assume that the temperature of the object does not change appreciably in the process. Express your answer numerically in joules per kelvin. ΔS = nothing J/K Request Answer Part F An object at 400 K absorbs 25.0 kJ of heat from the surroundings. What is the change in entropy ΔS of the object? Assume that the temperature of the object does not change appreciably in the process. Express your answer numerically in joules per kelvin. ΔS = nothing J/K Request Answer Part G Two objects form a closed system. One object, which is at 400 K, absorbs 25.0 kJ of heat from the other object, which is at 500 K. What is the net change in entropy ΔSsys of the system? Assume that the temperatures of the objects do not change appreciably in the process. Express your answer numerically in joules per kelvin.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2


Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1
You know the right answer?
An object at 20∘C absorbs 25.0 J of heat. What is the change in entropy ΔS of the object? Express yo...
Questions




Mathematics, 03.12.2021 14:00

Mathematics, 03.12.2021 14:00

English, 03.12.2021 14:00

Spanish, 03.12.2021 14:00

Mathematics, 03.12.2021 14:00



English, 03.12.2021 14:00


Advanced Placement (AP), 03.12.2021 14:00


English, 03.12.2021 14:00




English, 03.12.2021 14:00