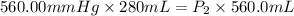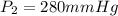Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3
You know the right answer?
A sample of gas occupies 280 mL when the pressure is 560.00 mm Hg . If the temperature remains const...
Questions


German, 03.12.2020 01:50


Mathematics, 03.12.2020 01:50



Engineering, 03.12.2020 01:50



Biology, 03.12.2020 01:50

Mathematics, 03.12.2020 01:50

History, 03.12.2020 01:50

Physics, 03.12.2020 01:50

Health, 03.12.2020 01:50


Computers and Technology, 03.12.2020 01:50


Spanish, 03.12.2020 01:50

History, 03.12.2020 01:50

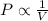
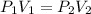
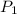 = initial pressure = 560.00 mmHg
= initial pressure = 560.00 mmHg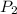 = final pressure = ?
= final pressure = ?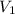 = initial volume = 280 mL
= initial volume = 280 mL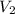 = final volume = 560.0 mL
= final volume = 560.0 mL