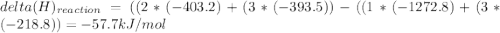Chemistry, 05.05.2020 08:30 jackiediaz
Use the given heats of formation to calculate the enthalpy change for this reaction. B2O3(g) + 3COCl2(g) →2BCl3(g) + 3CO2(g) ΔHof of B2O3(g) is –1272.8 kJ∙mol–1 ΔHof of BCl3(g) is –403.8 kJ∙mol–1 ΔHof of COCl2(g) is –218.8 kJ∙mol–1 ΔHof of CO2(g) is –393.5 kJ∙mol–1 Question 8 options: 1) 649.3 kJ·mol–1 2) 354.9 kJ·mol–1 3) –58.9 kJ·mol–1 4) –3917.3 kJ·mol–1

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 23.06.2019 00:00
Exit what is the density of an object having a mass of 5.0 g and a volume of 45.0 cm3?
Answers: 1
You know the right answer?
Use the given heats of formation to calculate the enthalpy change for this reaction. B2O3(g) + 3COCl...
Questions

Mathematics, 27.05.2021 19:20


Mathematics, 27.05.2021 19:20


Mathematics, 27.05.2021 19:20

Geography, 27.05.2021 19:20

Mathematics, 27.05.2021 19:20

Chemistry, 27.05.2021 19:20


History, 27.05.2021 19:20

Mathematics, 27.05.2021 19:20



Mathematics, 27.05.2021 19:20


Mathematics, 27.05.2021 19:20

Mathematics, 27.05.2021 19:20

Engineering, 27.05.2021 19:20


History, 27.05.2021 19:20