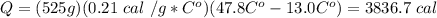How many calories of heat are required to raise the temperature of 525g of
Aluminum from 13.0°...

Chemistry, 05.05.2020 15:20 heyperdomo4369
How many calories of heat are required to raise the temperature of 525g of
Aluminum from 13.0°C to 47.8°C? (CAL= 0.21 cal/g°C)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

You know the right answer?
Questions


History, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00


Chemistry, 13.12.2020 14:00



History, 13.12.2020 14:00

English, 13.12.2020 14:00

Social Studies, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00

English, 13.12.2020 14:00



History, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00

Computers and Technology, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00