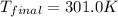Chemistry, 05.05.2020 15:04 BeautyxQueen
A 31.5 g wafer of pure gold initially at 69.4 ∘C is submerged into 63.4 g of water at 27.4 ∘C in an insulated container.
What is the final temperature of both substances at thermal equilibrium?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
What does earth’s rotation on its axis cause? the tides night and day passing of years phases of the moon
Answers: 1

Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 23.06.2019 00:00
If many scientists conduct the same or similar experiments, and all obtain similar results, a can be written, which is a generally agreed-upon statement that explains and predicts how a natural phenomenon works.
Answers: 1
You know the right answer?
A 31.5 g wafer of pure gold initially at 69.4 ∘C is submerged into 63.4 g of water at 27.4 ∘C in an...
Questions



Biology, 26.07.2019 18:30


English, 26.07.2019 18:30





Arts, 26.07.2019 18:30

Arts, 26.07.2019 18:30

Arts, 26.07.2019 18:30


Biology, 26.07.2019 18:30

Biology, 26.07.2019 18:30

Biology, 26.07.2019 18:30

Business, 26.07.2019 18:30



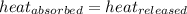
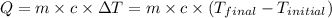
![m_1\times c_1\times (T_{final}-T_1)=-[m_2\times c_2\times (T_{final}-T_2)]](/tpl/images/0639/3665/09236.png) .................(1)
.................(1)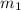 = mass of gold = 31.5 g
= mass of gold = 31.5 g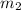 = mass of water = 63.4 g
= mass of water = 63.4 g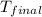 = final temperature = ?
= final temperature = ?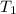 = temperature of gold =
= temperature of gold = 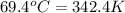
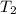 = temperature of water =
= temperature of water = 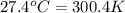
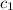 = specific heat of gold =
= specific heat of gold = 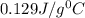
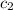 = specific heat of water=
= specific heat of water= 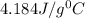
![-31.5\times 0.129\times (T_{final}-342.4)=[63.4\times 4.184\times (T_{final}-300.4)]](/tpl/images/0639/3665/eb953.png)