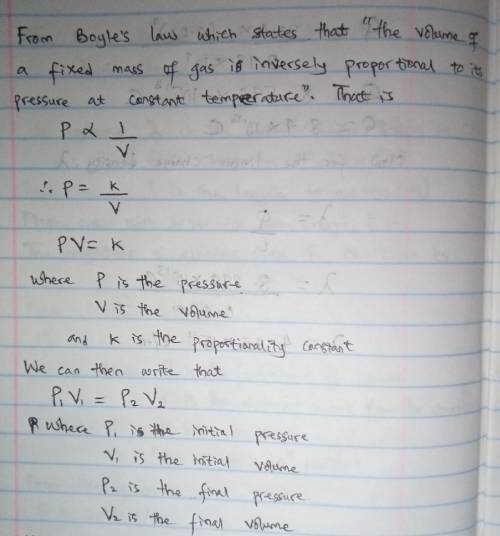Chemistry, 05.05.2020 20:06 Rosy1234Locks
The stopcock connecting a 1.30 L bulb containing xenon gas at a pressure of 6.15 atm, and a 4.25 L bulb containing oxygen gas at a pressure of 3.53 atm, is opened and the gases are allowed to mix. Assuming that the temperature remains constant, the final pressure in the system is 8.33 atm. True or false?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What royal scientist used the 29th day of frozen vapor to encounter elements for mastering new culinary creations?
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2
You know the right answer?
The stopcock connecting a 1.30 L bulb containing xenon gas at a pressure of 6.15 atm, and a 4.25 L b...
Questions


English, 11.11.2019 07:31


Mathematics, 11.11.2019 07:31

Mathematics, 11.11.2019 07:31


Mathematics, 11.11.2019 07:31


Mathematics, 11.11.2019 07:31

Health, 11.11.2019 07:31


Mathematics, 11.11.2019 07:31

English, 11.11.2019 07:31

Mathematics, 11.11.2019 07:31


Mathematics, 11.11.2019 07:31

Mathematics, 11.11.2019 07:31


Biology, 11.11.2019 07:31