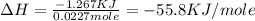When 22.7 mL of 0.500 M H2SO4 is added to 22.7 mL of 1.00 M KOH in a coffee-cup calorimeter at 23.50°C, the temperature rises to 30.17°C. Calculate ΔH of this reaction. (Assume that the total volume is the sum of the individual volumes and that the density and specific heat capacity of the solution are the same as for pure water.) (d for water = 1.00 g/mL; c for water = 4.184 J/g·°C.)___kJ/mol H2O

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 04:00
Why must humans find substitutes for many minerals found on earth? (a) form at an extremely slow rate (b) controlled by other countries (c) too deep in the earth to collect
Answers: 1

Chemistry, 23.06.2019 10:20
El amoniaco y el fluor reaccionan para formar tetrafluoruro de dinitrogeno y fluoruro de hidrogeno. segun la reaccion: nh3 + f2 ⇒ n2f4 + hf si reaccionan 5 gramos de amoniaco y 20 gramos de fuor, ¿cuantos gramos de fluoruro de hidrogeno se producen?
Answers: 2

Chemistry, 23.06.2019 10:30
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 2

Chemistry, 23.06.2019 23:00
In the chemical formula 3h2 identify the subscript identify the coefficient how many atoms of hydrogen are present
Answers: 3
You know the right answer?
When 22.7 mL of 0.500 M H2SO4 is added to 22.7 mL of 1.00 M KOH in a coffee-cup calorimeter at 23.50...
Questions


Mathematics, 13.01.2021 22:30


History, 13.01.2021 22:30

Mathematics, 13.01.2021 22:30

Arts, 13.01.2021 22:30


History, 13.01.2021 22:30

Physics, 13.01.2021 22:30




Mathematics, 13.01.2021 22:30


Mathematics, 13.01.2021 22:30


English, 13.01.2021 22:30

Mathematics, 13.01.2021 22:30

Mathematics, 13.01.2021 22:30

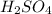 and KOH.
and KOH.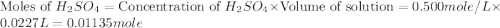
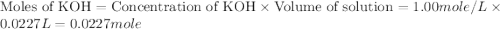
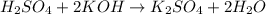
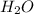
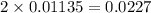 moles of
moles of 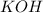 react to give 2 moles of
react to give 2 moles of 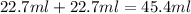
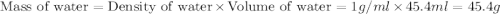
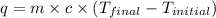
 = specific heat of water =
= specific heat of water = 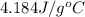
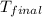 = final temperature of water =
= final temperature of water = 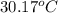
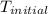 = initial temperature of water =
= initial temperature of water = 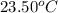
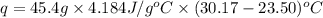
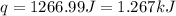
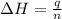
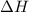 = enthalpy of neutralization = ?
= enthalpy of neutralization = ?