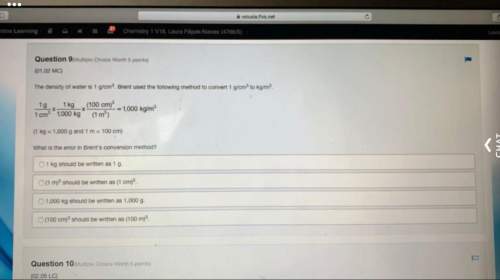
Chemistry, 27.04.2020 02:13 llewis2731
Given that the molar mass of NaNO3 is 85.00 g/mol, what mass of NaNO3 is needed to make 4.50 L of a 1.50 M NaNO3
solution?
moles of solute
Use molarity
liters of solution
6.75 g
18.99
255 g
574 g

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 21:30
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2

Chemistry, 23.06.2019 00:30
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1


Chemistry, 23.06.2019 13:20
Aluminum reacts with sulfuric acid to produce aluminum sulfate and hydrogen gas. how many grams of aluminum sulfate would be formed if 250 g h 2 so 4 completely reacted with aluminum? 2al( s ) + 3h 2 so 4 ( aq ) ? al 2 (so 4 ) 3 ( aq ) + 3h 2 ( g )
Answers: 1
You know the right answer?
Given that the molar mass of NaNO3 is 85.00 g/mol, what mass of NaNO3 is needed to make 4.50 L of a...
Questions

Mathematics, 21.04.2020 13:40

Social Studies, 21.04.2020 13:40

Mathematics, 21.04.2020 13:40

History, 21.04.2020 13:41

English, 21.04.2020 13:43

Physics, 21.04.2020 13:43



English, 21.04.2020 13:45




Biology, 21.04.2020 13:55

Mathematics, 21.04.2020 13:55


Mathematics, 21.04.2020 13:55


Arts, 21.04.2020 13:55


Mathematics, 21.04.2020 13:55