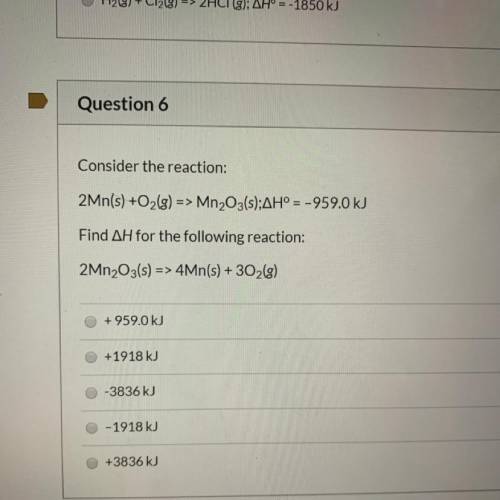
Consider the reaction:
2Mn(s) +O2(g) => Mn2O3(s);AH° = -959.0 kJ
Find AH for the foll...

Chemistry, 27.04.2020 01:29 CatsandDogsaredabest
Consider the reaction:
2Mn(s) +O2(g) => Mn2O3(s);AH° = -959.0 kJ
Find AH for the following reaction:
2Mn2O3(s) => 4Mn(s) + 302(g)
+959.0 kJ
+1918 kJ
-3836 kJ
O-1918 kJ
+3836 kJ

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3
You know the right answer?
Questions

Mathematics, 20.07.2019 13:00





History, 20.07.2019 13:00

Mathematics, 20.07.2019 13:00


Mathematics, 20.07.2019 13:00

Mathematics, 20.07.2019 13:00


Mathematics, 20.07.2019 13:00


Mathematics, 20.07.2019 13:00


Health, 20.07.2019 13:00

Mathematics, 20.07.2019 13:00






