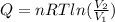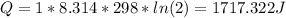Chemistry, 25.04.2020 09:15 katherineweightman
Calculate the heat transferred to the system when 1.00 mol of a perfect gas expands reversibly at a
Constant temperature of 25°C so that its volume doubles.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1


Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
You know the right answer?
Calculate the heat transferred to the system when 1.00 mol of a perfect gas expands reversibly at a<...
Questions

Mathematics, 24.10.2019 10:43


Biology, 24.10.2019 10:43







Mathematics, 24.10.2019 10:43

History, 24.10.2019 10:43

World Languages, 24.10.2019 10:43


History, 24.10.2019 10:43






Health, 24.10.2019 10:43

 (volume doubles due the gas expasion)
(volume doubles due the gas expasion)