Chemistry, 25.04.2020 04:09 hellodarkness14
A piece of copper (12.0 g) is heated to 100.0 °C. A piece of chromium (also 12.0 g) is chilled in an ice bath to 0 °C. The specific heat capacity of water is 4.184 J/g ⋅°C.
Both pieces of metal are placed in a beaker containing 200.0 g at 20.0 °C.
(a) Will the temperature of the water be greater than or less than 20.0 °C when thermal equilibrium is reached?
(b) Both pieces of metal are placed in a beaker containing 200.0 g at 20.0 °C. Calculate the final temperature.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
A piece of copper (12.0 g) is heated to 100.0 °C. A piece of chromium (also 12.0 g) is chilled in an...
Questions


English, 18.03.2021 01:00

Spanish, 18.03.2021 01:00


Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00




Mathematics, 18.03.2021 01:00

English, 18.03.2021 01:00

English, 18.03.2021 01:00



World Languages, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

English, 18.03.2021 01:00

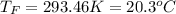
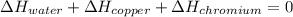
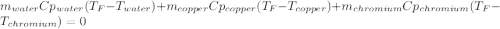 In such a way, by knowing that the heat capacities of copper and chromium are 0.386 and 0.45 J/(g°C) respectively, by solving for the equilibrium temperature one has:
In such a way, by knowing that the heat capacities of copper and chromium are 0.386 and 0.45 J/(g°C) respectively, by solving for the equilibrium temperature one has: