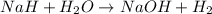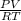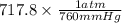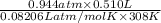Binary compounds of alkali metals and hydrogen react with water to liberate hydrogen gas. The hydrogen gas from the reaction of a sample of sodium hydride with an excess of water fills a volume of 0.510 L above the water. The temperature of the gas is 35 ∘C and the total pressure is 760 mmHg. Find the mass of H2 liberated and the mass of NaH that reacted.

Answers: 3


Another question on Chemistry


Chemistry, 23.06.2019 00:30
Ok, so i have 2 questions. try to answer them both: (the topic is fire) 1) how can you represent the chemical reaction of fire? 2) what kind of bond is formed in this chemical reaction
Answers: 3

Chemistry, 23.06.2019 11:20
The chemical composition of soil varies with depth. an article in communications in soil science and plant analysis describes chemical analyses of soil taken from a farm in western australia. fifty specimens were each taken at depths 50 and 250 cm. at a depth of 50 cm, the average no3 concentration (in mg/l) was 88.5 with a standard deviation of 49.4. at a depth of 250 cm, the average concentration was 110.6 with a standard deviation of 51.5. find a 95% confidence interval for the difference in no3 concentrations at the two depths.
Answers: 1

Chemistry, 23.06.2019 13:30
Determine the osmotic pressure at 25 °c of an aqueous solution that is 0.028 m nano3. a) 0.685 atm b) 0.0729 atm c) 1.37 atm d) 0.0364 atm e) 2.06 atm
Answers: 2
You know the right answer?
Binary compounds of alkali metals and hydrogen react with water to liberate hydrogen gas. The hydrog...
Questions




Mathematics, 04.07.2019 14:00

History, 04.07.2019 14:00

Mathematics, 04.07.2019 14:00



Biology, 04.07.2019 14:00

Mathematics, 04.07.2019 14:00

Social Studies, 04.07.2019 14:00

History, 04.07.2019 14:00

Social Studies, 04.07.2019 14:00

History, 04.07.2019 14:00


Social Studies, 04.07.2019 14:00

Mathematics, 04.07.2019 14:00


History, 04.07.2019 14:00

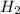 liberated is 0.0383 g and the mass of NaH that reacted 0.455 g.
liberated is 0.0383 g and the mass of NaH that reacted 0.455 g.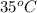 = (35 + 273) K = 308 K,
= (35 + 273) K = 308 K,