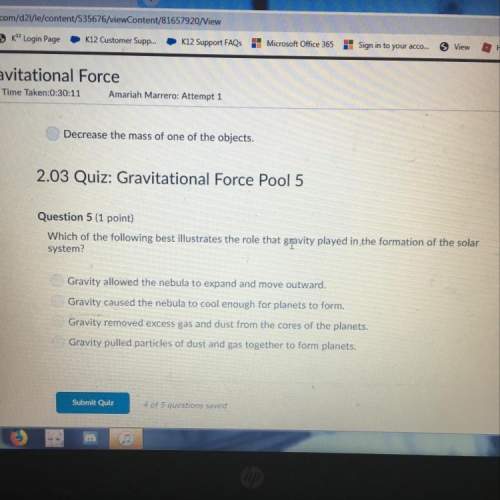
Chemistry, 25.04.2020 02:50 kattydominguez15
Consider the following electron configurations to answer the question: (i) 1s2 2s2 2p6 3s1 (ii) 1s2 2s2 2p6 3s2 (iii) 1s2 2s2 2p6 3s2 3p1 (iv) 1s2 2s2 2p6 3s2 3p4 (v) 1s2 2s2 2p6 3s2 3p5 The electron configuration that belongs to the atom with the lowest second ionization energy is

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
Consider the following electron configurations to answer the question: (i) 1s2 2s2 2p6 3s1 (ii) 1s2...
Questions


Mathematics, 06.08.2019 02:20



Advanced Placement (AP), 06.08.2019 02:20

History, 06.08.2019 02:20

English, 06.08.2019 02:20



Biology, 06.08.2019 02:20

Mathematics, 06.08.2019 02:20







Computers and Technology, 06.08.2019 02:20