
Chemistry, 25.04.2020 03:04 guapotaco8102
Consider two equal-volume flasks of gas at the same temperature and pressure. One gas, oxygen, has a molecular mass of 32. The other gas, nitrogen, has a molecular mass of 28. What is the ratio of the number of oxygen molecules to the number of nitrogen molecules in these flasks

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon? a balloon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1
You know the right answer?
Consider two equal-volume flasks of gas at the same temperature and pressure. One gas, oxygen, has a...
Questions

Biology, 08.07.2019 22:50


Physics, 08.07.2019 22:50


Advanced Placement (AP), 08.07.2019 22:50



Mathematics, 08.07.2019 22:50



Spanish, 08.07.2019 22:50



Health, 08.07.2019 22:50




Mathematics, 08.07.2019 22:50

Mathematics, 08.07.2019 22:50

Mathematics, 08.07.2019 22:50

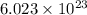 of particles.
of particles.

