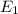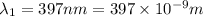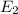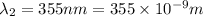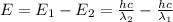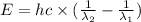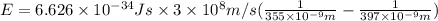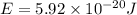Chemistry, 23.04.2020 04:10 iliketurtures
18-31. In formaldehyde, the transition n S p*(T1) occurs at 397 nm, and the n S p*(S1) transition comes at 355 nm. What is the differ- ence in energy (kJ/mol) between the S1 and T1 states? This differ- ence is due to the different electron spins in the two states.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2


Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
You know the right answer?
18-31. In formaldehyde, the transition n S p*(T1) occurs at 397 nm, and the n S p*(S1) transition co...
Questions


Biology, 08.12.2020 03:20

Arts, 08.12.2020 03:20

Business, 08.12.2020 03:20

English, 08.12.2020 03:20

English, 08.12.2020 03:20


English, 08.12.2020 03:20


Mathematics, 08.12.2020 03:20





Mathematics, 08.12.2020 03:20


Mathematics, 08.12.2020 03:20

Mathematics, 08.12.2020 03:20

Mathematics, 08.12.2020 03:20

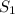 and
and 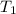 states is 35.7 kJ/mol.
states is 35.7 kJ/mol.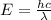
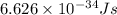
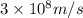
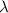 = Wavelength of the electromagnetic radiations.
= Wavelength of the electromagnetic radiations.