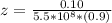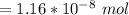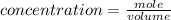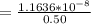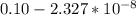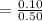Chemistry, 23.04.2020 01:17 gildedav001
A solution is prepared by adding 0.10 mole of Ni(NH3)6Cl2 to 0.50 L of 3.0 M NH3. Calculate [Ni(NH3)62 ] and [Ni2 ] in this solution. Koverall for Ni(NH3)62 is 5.5 x 108. That is, 5.5 x 108

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1
You know the right answer?
A solution is prepared by adding 0.10 mole of Ni(NH3)6Cl2 to 0.50 L of 3.0 M NH3. Calculate [Ni(NH3)...
Questions



Advanced Placement (AP), 25.02.2021 01:30

Social Studies, 25.02.2021 01:30


Mathematics, 25.02.2021 01:30



Arts, 25.02.2021 01:30

Mathematics, 25.02.2021 01:30

Mathematics, 25.02.2021 01:30


Chemistry, 25.02.2021 01:30

Chemistry, 25.02.2021 01:30

Mathematics, 25.02.2021 01:30

Geography, 25.02.2021 01:30


Mathematics, 25.02.2021 01:30


Mathematics, 25.02.2021 01:30

![[Ni^{2+}]= 2.327*10^{-8} M](/tpl/images/0620/1201/754f2.png)
![[Ni(NH_3)_6]^{2+} _{(aq)}]= 0.3 M](/tpl/images/0620/1201/266ad.png)
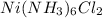 is
is 
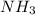 is =
is = 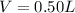
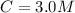
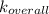 for
for ![[Ni(NH_3) _6^{2+}]](/tpl/images/0620/1201/584c3.png) is
is 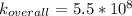
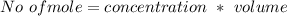
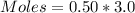
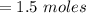
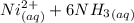 ⇄
⇄ ![[Ni(NH_3)_6]^{2+} _{(aq)}](/tpl/images/0620/1201/fd544.png)
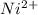
![0.10moles [Ni ^{2+}] * \frac{6moles NH_3}{1 mole [N_i^{2+}]}](/tpl/images/0620/1201/88989.png)
![= 0.6 moles [Ni^{2+}]](/tpl/images/0620/1201/c903d.png)
![1.5 moles [NH_3] * \frac{6moles NH_3}{1 mole [N_i^{2+}]}](/tpl/images/0620/1201/a0abd.png)
![= 9 moles [NH_3]](/tpl/images/0620/1201/cec0c.png)
![0.10 [Ni^{2+}] *\frac{1 moles [Ni(NH_3)_6]^{2+}}{1 mol [Ni^{2+}]}](/tpl/images/0620/1201/cfb14.png)
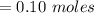
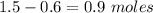
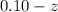
![K_{overall} = \frac{[Ni (NH_3) _6]^{2+}}{[Ni^{2+} [NH_3] ^6]}](/tpl/images/0620/1201/c7d6a.png)
![5.5*10^{8} = \frac{0.10 - z}{[z] [0.9 + 6z]^6}](/tpl/images/0620/1201/31e23.png)
![5.5*10^{8} = \frac{0.10}{[z] [0.5]^6}](/tpl/images/0620/1201/7e13e.png)