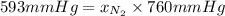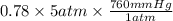Chemistry, 22.04.2020 23:16 juanitarodrigue
The partial pressure of N2 in the air is 593 mm Hg at 1 atm. What is the partial pressure of N2 in a bubble of air a scuba diver breathes when he is 132 ft below the surface of the water where the pressure is 5.00 atm?

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 23:50
How will the emission of an alpha particle affect the atomic number of an atom
Answers: 3

Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
You know the right answer?
The partial pressure of N2 in the air is 593 mm Hg at 1 atm. What is the partial pressure of N2 in a...
Questions

History, 30.06.2019 16:00

English, 30.06.2019 16:00

Biology, 30.06.2019 16:00

Mathematics, 30.06.2019 16:00


Health, 30.06.2019 16:00

History, 30.06.2019 16:00

Mathematics, 30.06.2019 16:00


Physics, 30.06.2019 16:00

Biology, 30.06.2019 16:00


Physics, 30.06.2019 16:00

Geography, 30.06.2019 16:00

Mathematics, 30.06.2019 16:00


History, 30.06.2019 16:00

Chemistry, 30.06.2019 16:00


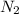 at a depth of 132 ft below sea level is 2964 mm Hg.
at a depth of 132 ft below sea level is 2964 mm Hg.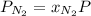
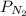 = partial pressure of
= partial pressure of 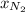 = mole fraction of
= mole fraction of