The reaction of hydrochloric acid (HCl) with ammonia (NH3) is described by the equation:
...

The reaction of hydrochloric acid (HCl) with ammonia (NH3) is described by the equation:
HCl + NH3 → NH4Cl
A student is titrating 50 mL of 0.32 M NH3 with 0.5 M HCl. How much hydrochloric acid must be added to react completely with the ammonia?
A. 6.4 mL
B. 16.0 mL
C. 32.0 mL
D. 50.0 mL

Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1

Chemistry, 23.06.2019 05:00
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2

Chemistry, 23.06.2019 08:30
Kelly has come up with an explanation for why her sister is sometimes in a good mood and other times in a bad mood. she speculates that it is based on the hours of sleep her sister got the previous night. this explanation for her sister's behaviors is an example of a(n)
Answers: 3
You know the right answer?
Questions

Mathematics, 01.06.2021 18:50

Mathematics, 01.06.2021 18:50




Mathematics, 01.06.2021 18:50

Mathematics, 01.06.2021 18:50




Social Studies, 01.06.2021 18:50





Mathematics, 01.06.2021 18:50

Mathematics, 01.06.2021 18:50



 are the initial molarity and volume of NH₃.
are the initial molarity and volume of NH₃.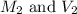 are the final molarity and volume of HCl.
are the final molarity and volume of HCl.